Based on Moore, V. and Quigley, M., ‘Open-Source Diabetes Technologies: On Clinician Liability and Patient Safety‘, published in Patient Safety Law, Ethics and Practice in the NHS (Routledge, 2026). In previous work we examined how GMC guidance on prescribing and consent applies to clinicians caring for OS AID users, concluding that nothing in it should … Continue reading “Can Clinicians Be Held Liable for Caring for OS AID Users?”
Category: DIY APS
Do-It-Yourself Diabetes Technology and the Law
This piece was originally posted as a Birmingham Law School Research Blog Post This post summarises a paper we published a few months ago in Diabetic Medicine: ‘#WeAreNotWaiting DIY Artificial Pancreas Systems and the Challenges for the Law’* Authors: Rachael Dickson, Jessica Bell, Amber Dar, Laura Downey, Victoria Moore, Muireann Quigley Post written by: Rachael Dickson OS AID … Continue reading “Do-It-Yourself Diabetes Technology and the Law”
Prescribing Unapproved Medical Devices? The Case of DIY Artificial Pancreas Systems
This post is a summary of our forthcoming paper in Medical Law International ‘Prescribing Unapproved Medical Devices? The Case of DIY Artificial Pancreas Systems’ Authors: Joseph T F Roberts, Victoria Moore, Muireann Quigley Healthcare technology innovation in type 1 diabetes (T1D) management has until recently been a relatively slow process. Tired of waiting for commercial manufacturers … Continue reading “Prescribing Unapproved Medical Devices? The Case of DIY Artificial Pancreas Systems”
Prescribing and DIY Artificial Pancreas Systems
In this post we consider how clinicians can follow the standards set by the General Medical Council when making prescribing decisions for people who are using or considering using DIY APS. In DIY APS, each component of the system that a doctor is responsible for prescribing (insulin, CGM, and insulin pump) are appropriately licensed or … Continue reading “Prescribing and DIY Artificial Pancreas Systems”
Now that we have commercial Artificial Pancreas Systems, is there still a place for DIY APS?
Dr Rachael Dickson explores whether advances in market authorised artificial pancreas systems spell the end of DIY APS and resolve the legal uncertainty that these systems raise. Over the last decade the #WeAreNotWaiting community has come together to develop DIY Artificial Pancreas Systems to help them manage their diabetes. Frustrated by the lack of commercial … Continue reading “Now that we have commercial Artificial Pancreas Systems, is there still a place for DIY APS?”
Perspectives on DIY APS: Users, Healthcare Professionals, and Bioethicists
A number of groups have a stake in DIY Artificial Pancreas Systems (APS) technology including users, healthcare providers, manufacturers, and clinicians. Given their diverse goals, needs, and professional commitments, these different groups have different perspectives on DIY APS use. A recently published paper by Dr Jaclyn Shepard and colleagues reports the views expressed by stakeholders … Continue reading “Perspectives on DIY APS: Users, Healthcare Professionals, and Bioethicists”
The DIY artificial pancreas: Who is liable if something goes wrong?
In this post we sketch out different parts of the ‘looping’ ecosystem and start to consider the issue of liability if something should go wrong. In a series of posts, we have been looking at the use of DIY Artificial Pancreas Systems (DIY APS or ‘looping’). These systems – made up of an insulin pump, … Continue reading “The DIY artificial pancreas: Who is liable if something goes wrong?”
DIY Artificial Pancreas Systems: Safety concerns and potential risks in perspective?
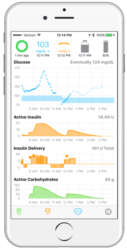
In this post we explore some of the risks and benefits of DIY Artificial Pancreas technologies. In future posts we will take a look at the law, ethics, and policy implications of these. In a previous post we explained how Do-it-Yourself Artificial Pancreas Systems (DIY APS) are systems made up of four interconnected components: a continuous … Continue reading “DIY Artificial Pancreas Systems: Safety concerns and potential risks in perspective?”
Patients are doing it for themselves: DIY artificial pancreas systems and the challenge for doctors
Some people with type 1 diabetes (T1D) find current treatment options to be inadequate and unsatisfactory and have taken it upon themselves to design technological solutions which work for them. Tired of waiting for commercial solutions that meet their needs, patients are constructing their own Do-it-Yourself Artificial Pancreas Systems (DIY APS). Although still confined to … Continue reading “Patients are doing it for themselves: DIY artificial pancreas systems and the challenge for doctors”